Difference between ammonia and ammonium often confuses students studying chemistry. Imagine a science class where a teacher mentions both words during a lesson about fertilizers and water chemistry.
At first, they sound almost identical. Yet ammonia and ammonium represent two very different chemical forms of nitrogen.
Understanding the difference between ammonia and ammonium helps students grasp how nitrogen behaves in soil, water, and living organisms.
When learners clearly understand the difference between ammonia and ammonium, they can better understand agriculture, environmental science, and chemical reactions. Let’s explore their differences step by step.
Key Difference Between the Two
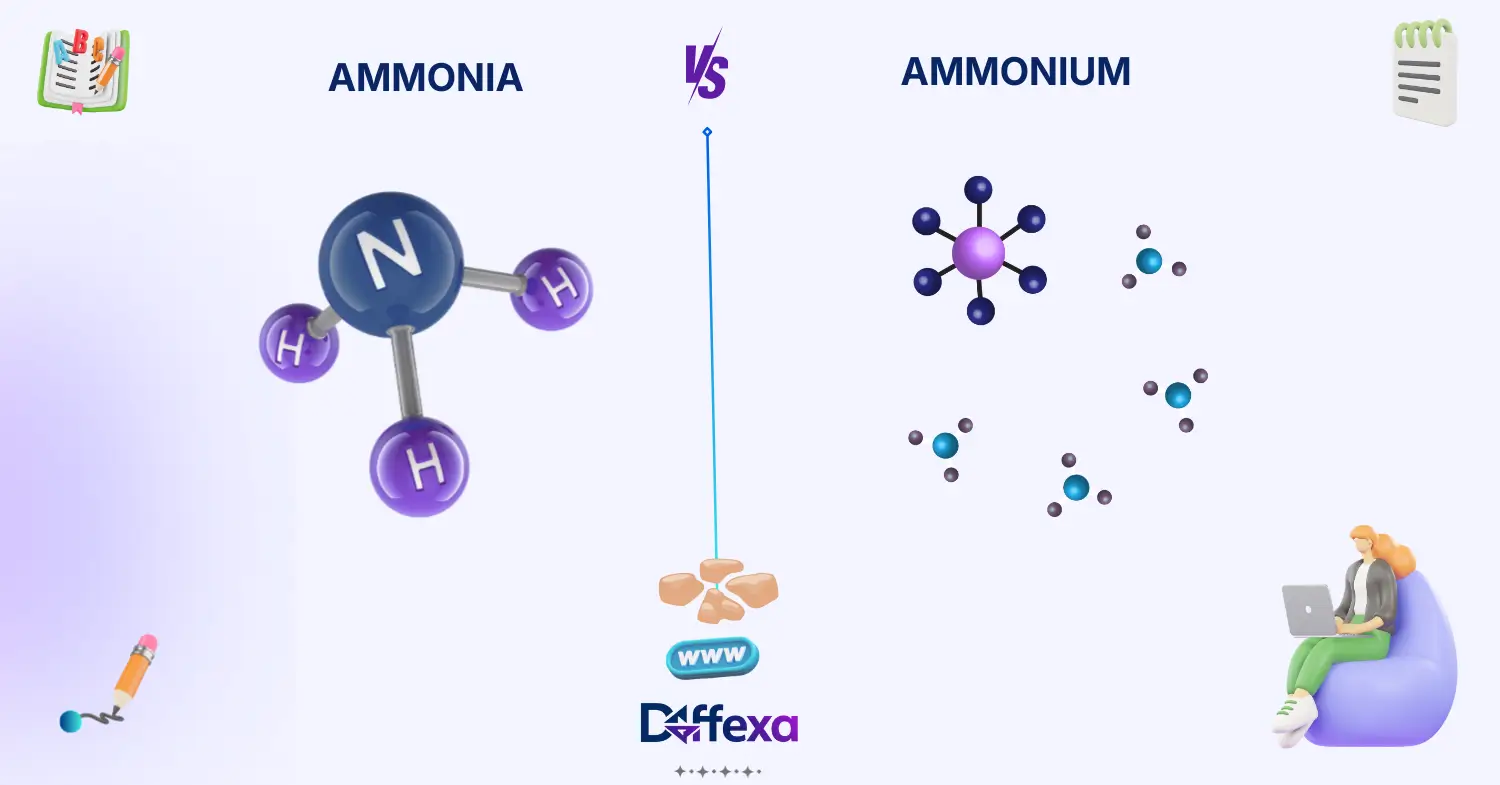
The main difference between ammonia and ammonium lies in their chemical structure and electrical charge.
Ammonia (NH₃) is a neutral molecule made of one nitrogen atom bonded to three hydrogen atoms.
Ammonium (NH₄⁺) is a positively charged ion formed when ammonia gains a hydrogen ion.
In simple terms:
- Ammonia = neutral gas molecule
- Ammonium = positively charged ion
Ammonia commonly exists as a gas with a strong smell. Ammonium usually appears dissolved in water or within chemical compounds.
Why Is Their Difference Necessary to Know
Understanding the difference between ammonia and ammonium matters in both education and real life.
Students studying chemistry, biology, or environmental science must know how these forms of nitrogen behave in reactions.
Professionals such as farmers, environmental scientists, and water treatment experts rely on this knowledge. The form of nitrogen present affects plant growth and water quality.
In society, ammonia and ammonium influence agriculture, pollution control, and ecosystem health. Knowing the difference helps people manage fertilizers and protect water systems.
Now let’s learn how these words are pronounced.
Pronunciation
Ammonia
US: /əˈmoʊ.ni.ə/
UK: /əˈməʊ.ni.ə/
Ammonium
US: /əˈmoʊ.ni.əm/
UK: /əˈməʊ.ni.əm/
The words sound similar, but their meanings in chemistry are quite different. Let’s examine their core definitions next.
Core Definitions
Ammonia
Ammonia is a colorless gas made of nitrogen and hydrogen with the formula NH₃. It has a strong smell and dissolves easily in water.
Scientists use ammonia in fertilizers, cleaning products, and industrial processes.
Example:
Example: Farmers sometimes use ammonia‑based fertilizers to improve soil nutrients.
Ammonium
Ammonium is a positively charged ion with the formula NH₄⁺. It forms when ammonia reacts with a hydrogen ion.
Ammonium usually exists dissolved in water or inside salts such as ammonium nitrate.
Example:
Example: Plants absorb nitrogen from soil in the form of ammonium ions.
10 Clear Differences Between Ammonia and Ammonium
1. Chemical Structure
Ammonia contains one nitrogen atom and three hydrogen atoms.
Ammonium contains one nitrogen atom and four hydrogen atoms.
Example for Ammonia:
Example: Ammonia gas leaks can be detected by their strong smell.
Example for Ammonium:
Example: Soil tests often measure ammonium levels.
2. Electrical Charge
Ammonia is electrically neutral.
Ammonium carries a positive charge.
Example for Ammonia:
Example: Ammonia molecules move freely in air.
Example for Ammonium:
Example: Ammonium ions interact with negatively charged particles.
3. Physical State
Ammonia commonly exists as a gas.
Ammonium usually exists in dissolved or solid forms.
Example for Ammonia:
Example: Ammonia gas is stored in pressurized tanks.
Example for Ammonium:
Example: Ammonium salts appear as white crystals.
4. Chemical Behavior
Ammonia acts as a weak base.
Ammonium behaves as a weak acid in some reactions.
Example for Ammonia:
Example: Ammonia reacts with acids to form salts.
Example for Ammonium:
Example: Ammonium releases hydrogen ions in solution.
5. Stability in Water
Ammonia easily dissolves in water.
Ammonium remains stable in aqueous solutions.
Example for Ammonia:
Example: Ammonia dissolves quickly in water tanks.
Example for Ammonium:
Example: Ammonium ions stay dissolved in soil moisture.
6. Smell
Ammonia has a strong, sharp odor.
Ammonium compounds usually have little smell.
Example for Ammonia:
Example: Household cleaners often smell like ammonia.
Example for Ammonium:
Example: Ammonium nitrate fertilizer has little odor.
7. Environmental Presence
Ammonia forms in animal waste and decomposition.
Ammonium forms in soil and water after chemical reactions.
Example for Ammonia:
Example: Livestock farms release ammonia gas.
Example for Ammonium:
Example: Soil microbes convert nitrogen into ammonium.
8. Role in Agriculture
Ammonia serves as a raw fertilizer chemical.
Ammonium acts as a plant nutrient.
Example for Ammonia:
Example: Ammonia injection fertilizes farmland.
Example for Ammonium:
Example: Plants absorb ammonium through roots.
9. Chemical Form
Ammonia is a molecule.
Ammonium is an ion.
Example for Ammonia:
Example: Chemists write ammonia as NH₃.
Example for Ammonium:
Example: Ammonium appears as NH₄⁺ in formulas.
10. Scientific Context
Ammonia appears in gas chemistry and industrial production.
Ammonium appears in environmental and biological chemistry.
Example for Ammonia:
Example: Industrial plants produce ammonia for fertilizers.
Example for Ammonium:
Example: Water scientists monitor ammonium levels.
Why Knowing the Difference Matters
Understanding the difference between ammonia and ammonium improves scientific knowledge and communication.
Students studying chemistry or biology must distinguish these two forms of nitrogen to understand reactions and environmental cycles.
Professionals such as farmers, environmental engineers, and chemists rely on this knowledge when managing soil nutrients or water treatment systems.
Real‑world consequences of confusion
Confusing ammonia with ammonium could lead to mistakes in fertilizer use or pollution control. Accurate understanding helps protect ecosystems and agricultural productivity.
Why People Get Confused
Similar Spelling
The words share almost identical spelling, which causes confusion.
Semantic Overlap
Both refer to nitrogen‑hydrogen compounds in chemistry.
Context‑Based Usage
In everyday speech, people sometimes use the terms interchangeably.
Informal Speech
Non‑scientific discussions often simplify the terminology.
Connotation & Emotional Tone
Connotation = emotional meaning attached to a word.
In chemistry terms, emotional tone is minimal. However, context may still influence perception.
Ammonia
Positive: Useful in agriculture and industry.
Example: Ammonia helps produce fertilizers that support food production.
Negative: Strong smell or potential toxicity.
Example: Ammonia leaks can irritate the eyes.
Neutral: Scientific description in chemistry.
Example: Ammonia is composed of nitrogen and hydrogen.
Ammonium
Positive: Important plant nutrient.
Example: Ammonium supports plant growth.
Negative: Excess ammonium can cause water pollution.
Example: High ammonium levels affect aquatic ecosystems.
Neutral: Common chemical ion in nature.
Example: Ammonium appears in many chemical reactions.
Usage in Metaphors, Similes & Idioms
These words rarely appear in idioms or metaphors. They mainly belong to scientific language.
Example sentence:
“Scientists measured ammonia levels in the air and ammonium levels in the soil.”
This comparison highlights their different forms.
Comparison Table
| Feature | Ammonia | Ammonium |
|---|---|---|
| Meaning | Neutral molecule NH₃ | Positive ion NH₄⁺ |
| Tone | Scientific | Scientific |
| Usage | Gas, industry | Soil and water chemistry |
| Context | Industrial processes | Biological and environmental systems |
| Formality | Scientific terminology | Scientific terminology |
Which Is Better in What Situation?
When to Use Ammonia
Use ammonia when referring to the gaseous chemical NH₃. It appears in industrial chemistry, fertilizers, and cleaning products.
When to Use Ammonium
Use ammonium when discussing the NH₄⁺ ion in water, soil, or chemical compounds.
Situational Clarity
Scientists use ammonia when describing gas reactions. They use ammonium when describing ions in solutions.
Contextual Correctness
Choosing the correct term ensures scientific accuracy in research, agriculture, and environmental studies.
Literary or Cultural References
Although these chemicals mainly appear in science, they sometimes appear in educational media.
Book:
Chemistry: The Central Science (Science Textbook, Brown, LeMay, 2018)
Movie:
The Martian (USA, 2015) features scientific discussions about chemistry and agriculture in extreme environments.
FAQs
1. Is ammonium the same as ammonia?
No. Ammonia is a neutral molecule (NH₃). Ammonium is a positively charged ion (NH₄⁺) formed when ammonia gains a hydrogen ion.
2. Which one do plants absorb?
Plants can absorb nitrogen as ammonium ions. Soil microbes convert nitrogen compounds into this form.
3. Why does ammonia smell strong?
Ammonia gas easily escapes into the air. Its chemical structure produces a sharp, noticeable odor.
4. Is ammonium harmful?
In moderate amounts, ammonium is useful for plants. However, excessive levels can pollute water and harm aquatic life.
5. How are ammonia and ammonium connected?
Ammonium forms when ammonia reacts with hydrogen ions. The two chemicals constantly convert between each other in water depending on pH.
Conclusion
The difference between ammonia and ammonium centers on chemical structure and electrical charge. Ammonia is a neutral molecule composed of nitrogen and hydrogen, while ammonium is a positively charged ion formed when ammonia gains a hydrogen ion.
Although the names sound similar, their behavior in chemistry differs greatly. Ammonia often exists as a gas, while ammonium usually appears dissolved in water or within chemical compounds.
Understanding this difference helps students, scientists, and professionals interpret chemical reactions and environmental processes correctly.
As you continue studying chemistry, recognizing subtle distinctions like this strengthens your scientific understanding and improves clear communication in science. 🧪
Sajid Ali is an English language educator and content specialist with 7+ years of teaching experience. He holds master’s degrees in Information Technology and Education from the Virtual University of Pakistan and writes SEO-optimized, learner-friendly vocabulary content for diffexa.com.